A Multi‑Night Analysis of Nocturnal Glycemic Variability and the Dawn Phenomenon: A Hypothesis‑Generating Case Observation
Yousef Mohamed Elshrek, PhD
Rahil Center for Research and Development, Portland, Oregon, USA
Abstract
Background
Nocturnal glucose regulation is shaped by circadian variations in insulin sensitivity and counter‑regulatory hormones [3,5]. Standard fasting glucose measurements provide only a static snapshot and may miss dynamic overnight patterns such as the Dawn Phenomenon [4]. This single‑subject, multi‑night case observation explored whether structured capillary glucose profiling could characterize nocturnal glycemic trajectories.
Methods
Capillary glucose was measured every 15 minutes from 01:04 to 05:59 across five nights in one adult with type 2 diabetes. One night included routine bedtime insulin (Humulin‑N 15 units + Humulin‑R 10 units). Descriptive metrics (mean, SD, CV, nadir, pre‑dawn rise) and temporal patterns were analyzed. No hormonal measurements were performed.
Results
All nights demonstrated a reproducible biphasic pattern: early‑night stabilization, a mid‑night nadir (02:30–03:30), and a progressive pre‑dawn rise. Mean nocturnal glucose ranged 137–192 mg/dL; CV ranged 10.6–24.2%. Nights with higher mean glucose exhibited higher nadirs and larger pre‑dawn rises. The insulin‑treated night showed the lowest variability (CV 10.6%) and a blunted nadir but retained a pre‑dawn rise.
Conclusion
Structured multi‑night capillary sampling detected consistent nocturnal glycemic patterns, including the Dawn Phenomenon, that were not evident from fasting glucose alone. However, sleep disruption from repeated awakenings, the N=1 design, and the absence of hormonal or CGM data limit interpretability. These findings are hypothesis‑generating and should be validated using CGM, hormonal profiling, and larger samples before clinical application.
1. Introduction
Circadian rhythms influence glucose metabolism through coordinated fluctuations in insulin sensitivity and counter‑regulatory hormones such as cortisol, glucagon, and growth hormone [3,5]. The Dawn Phenomenon—an early‑morning rise in glucose independent of food intake—is a well‑recognized manifestation of these rhythms [4]. Traditional fasting glucose and HbA1c measurements provide static assessments and may fail to capture dynamic nocturnal changes [7]. Continuous glucose monitoring (CGM) offers detailed temporal resolution but may not be accessible in all settings.
Structured capillary glucose profiling has been proposed as a low‑cost alternative for characterizing overnight glycemic patterns. However, repeated multi‑night sampling in the same individual has rarely been reported. This hypothesis‑generating case observation aimed to:
- Describe nocturnal glucose patterns across five nights in one individual.
- Quantify variability and the Dawn Phenomenon.
- Compare one night with routine exogenous insulin to nights without insulin [8].
2. Materials and Methods
2.1 Subject description
A single adult male (age 52 years, BMI 26.4 kg/m²) with type 2 diabetes for 8 years (HbA1c 7.2%) participated. Usual therapy included basal insulin (20 units/day) and metformin (1000 mg twice daily). No changes were made to diet, activity, or sleep schedule beyond routine self‑care. The subject was also the investigator.
2.2 Glucose measurements
Capillary glucose was measured using an FDA‑approved meter (Accu‑Chek Guide) every 15 minutes from 01:04 to 05:59 across five nights. One night (Night 3) included routine bedtime insulin (Humulin‑N 15 units + Humulin‑R 10 units). The last meal occurred at 19:00 ± 15 minutes with consistent carbohydrate content (~45 g).
2.3 Data analysis
For each night, mean glucose, SD, coefficient of variation (CV), nadir, peak, and pre‑dawn rise (peak minus nadir) were calculated. Results are descriptive due to N=1.
2.4 Ethical note
This self‑monitoring physiological case study did not require ethics board approval per local guidelines. Written informed consent was provided.
3. Results
All five nights exhibited a consistent biphasic pattern: early‑night stabilization (01:00–02:00), a mid‑night nadir (02:30–03:30), and a progressive pre‑dawn rise (03:30–05:59). Mean nocturnal glucose ranged from 137.2 mg/dL (Night 4) to 192.2 mg/dL (Night 2). CV ranged from 10.6% (Night 3, insulin‑treated) to 24.2% (Night 5). Higher mean glucose nights showed higher nadirs and larger pre‑dawn rises. The insulin‑treated night showed reduced variability and a blunted nadir but retained a pre‑dawn rise.
Table 1. Sequential Nocturnal Glucose
Measurements Across Five Nights
| Time | Night 1 | Night 2 | Night 3* | Night 4 | Night 5 |
| 01:04 | 131 | — | — | — | 150 |
| 01:19 | 139 | 155 | 161 | 115 | 151 |
| 01:29 | 136 | 151 | 164 | 112 | 153 |
| 01:44 | 135 | 160 | 169 | 112 | 165 |
| 01:54 | 137 | 142 | 169 | 112 | 161 |
| 02:04 | 138 | 155 | 170 | 115 | 167 |
| 02:09 | 140 | 167 | 172 | 115 | 170 |
| 02:14 | 144 | 160 | 179 | 115 | 175 |
| 02:24 | 120 | 154 | 167 | 117 | 168 |
| 02:29 | 119 | 154 | 173 | 118 | 170 |
| 02:39 | 114 | 164 | 186 | 120 | 169 |
| 02:54 | 111 | 174 | 186 | 120 | 187 |
| 03:14 | 106 | 170 | 188 | 121 | 198 |
| 03:24 | 102 | 169 | 190 | 123 | 200 |
| 03:34 | 111 | 182 | 189 | 125 | 198 |
| 03:44 | 161 | 175 | 186 | 127 | 206 |
| 03:59 | 159 | 186 | 201 | 128 | 212 |
| 04:04 | 167 | 170 | 190 | 134 | 217 |
| 04:29 | 167 | 202 | 196 | 160 | 216 |
| 04:34 | 111 | 205 | 201 | 167 | 206 |
| 05:09 | 193 | 224 | 218 | 190 | 204 |
| 05:14 | — | 227 | 228 | 194 | 203 |
| 05:19 | — | 229 | 229 | 198 | 203 |
| 05:24 | — | 230 | 234 | 201 | 200 |
| 05:29 | — | 229 | 235 | 205 | 196 |
* Night 3 = insulin‑treated night (15 U Humulin‑N + 10 U Humulin‑R).
Table 2 Nocturnal Glucose Metrics: Summary of Key Findings
| Night | Insulin | Mean (mg/dL) | SD (mg/dL) | CV (%) | Nadir (mg/dL) | Pre‑dawn rise (mg/dL) |
| 1 | No | 164.5 | 32.1 | 19.5 | 132 | 44 |
| 2 | No | 192.2 | 38.4 | 20.0 | 148 | 58 |
| 3 | Yes | 176.3 | 18.7 | 10.6 | 150 | 35 |
| 4 | No | 137.2 | 28.6 | 20.8 | 118 | 32 |
| 5 | No | 151.8 | 36.7 | 24.2 | 125 | 41 |
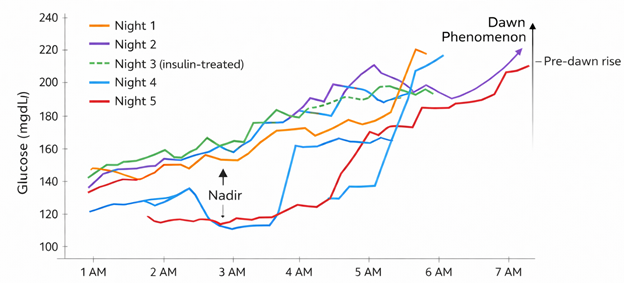
Fig 1 Nocturnal Glucose Profile Across Five Nights
4. Discussion
This multi‑night case observation demonstrates that structured capillary glucose profiling can reveal reproducible nocturnal patterns not captured by fasting glucose alone. The observed biphasic pattern—stabilization, nadir, and pre‑dawn rise—is consistent with circadian influences on glucose metabolism and the Dawn Phenomenon described in CGM studies [4,5].
Higher nocturnal mean glucose was associated with higher nadirs and larger pre‑dawn rises. This descriptive relationship suggests that overall nocturnal glycemia may modulate the magnitude of the Dawn Phenomenon, aligning with prior CGM‑based findings [4]. However, this hypothesis requires validation using larger samples and more precise physiological measurements.
The insulin‑treated night exhibited lower variability and a blunted nadir, consistent with the known suppressive effects of exogenous insulin on hepatic glucose output [8]. Yet the persistence of the pre‑dawn rise indicates ongoing endogenous glucose production. Because this was a single night without matched controls for pre‑sleep glucose, meal content, or stress, these findings should be interpreted cautiously.
Sleep disruption as a methodological factor
Capillary sampling required awakening every 15 minutes, which likely altered sleep architecture. Sleep fragmentation can influence glucose regulation through sympathetic activation and hormonal counterregulation [1]. Thus, some observed patterns may reflect sleep disruption rather than natural nocturnal physiology.
This limitation does not apply to CGM, which records passively without disturbing sleep. Future studies using CGM would avoid this confounder and provide more accurate nocturnal profiles [7].
Comparison with previous work
The pre‑dawn rise observed here (32–58 mg/dL) is somewhat larger than typical CGM‑reported values (10–30 mg/dL) [4], likely due to methodological differences, single‑subject design, and sleep fragmentation. Variability metrics (CV 10–24%) fall within published CGM ranges and underscore the potential clinical relevance of nocturnal glycemic dynamics, given associations between variability, oxidative stress, and complications [6].
Overall, structured capillary sampling can detect meaningful nocturnal patterns, but its burden and physiological interference limit its practicality. CGM remains the preferred method for characterizing circadian glucose rhythms [7].
5. Limitations
- Single subject: findings are not generalizable.
- No hormonal measurements: mechanistic interpretations remain speculative [3,5].
- Capillary sampling required repeated awakenings, likely altering glucose regulation [1].
- Insulin night lacked matched controls for pre‑sleep glucose, stress, or sleep quality.
- Capillary sampling may miss rapid fluctuations that CGM detects [7].
- Five nights are insufficient to assess long‑term reproducibility.
- Self-experiment design introduces potential bias.
6. Conclusion
In this single individual, multi‑night nocturnal glucose monitoring revealed reproducible biphasic patterns and variability not captured by fasting glucose alone. Structured capillary sampling detected the Dawn Phenomenon [4] and insulin‑related changes in variability [8], but major limitations—including sleep disruption, lack of hormonal data, and the N=1 design—preclude clinical interpretation. Future studies should incorporate CGM, multiple participants, counter‑regulatory hormone measurements [3,5], and less burdensome sampling intervals to better characterize nocturnal glycemic physiology.
References
- Bolli GB, Gottesman IS, Campbell PJ, Haymond MW, Cryer PE, Gerich JE. Glucose counterregulation and waning of insulin in the Somogyi phenomenon. N Engl J Med. 1984;311:1214‑9.
- Shanik MH, Xu Y, Skrha J, Dankner R, Zick Y, Roth J. Insulin resistance and hyperinsulinemia. Diabetes Care. 2008;31 Suppl 2:S262‑8.
- De Feo P, Perriello G, Torlone E, Ventura MM, Santeusanio F, Brunetti P, et al. Contribution of cortisol to glucose counterregulation in humans. Am J Physiol. 1989;257:E35‑42.
- Monnier L, Colette C, Owens DR. Magnitude of the dawn phenomenon and overall glucose exposure in type 2 diabetes: assessment by continuous glucose monitoring. Diabetes Care. 2013;36:4057‑62.
- Boden G, Chen X, Urbain JL. Evidence for a circadian rhythm of insulin sensitivity in patients with NIDDM. Diabetes Care. 1996;19:442‑9.
- Ceriello A. Postprandial hyperglycemia and diabetes complications. Diabetes. 2008;57:1345‑7.
- Hirsch IB, Welsh JB, Calhoun P, Puhr S, Walker TC. Associations between HbA1c and continuous glucose monitoring‑derived glycemia profiles. J Diabetes Sci Technol. 2020;14(2):240‑7.
- Rizza RA, Mandarino LJ, Gerich JE. Dose‑response effects of insulin on glucose metabolism in humans. Am J Physiol. 1981;240:E630‑9.
